Diverticulitis is an inflammatory process arising when one or more diverticula—small outpouchings in the colon wall—become inflamed or infected. While diverticulosis (pouches present) is common and often silent, diverticulitis can cause intense lower-abdominal pain, fever, and bowel-habit changes. This guide explains what it is, who gets it, how clinicians diagnose and stage it, and what treatments and day-to-day strategies can help you recover and reduce future flares.
What is diverticulitis?
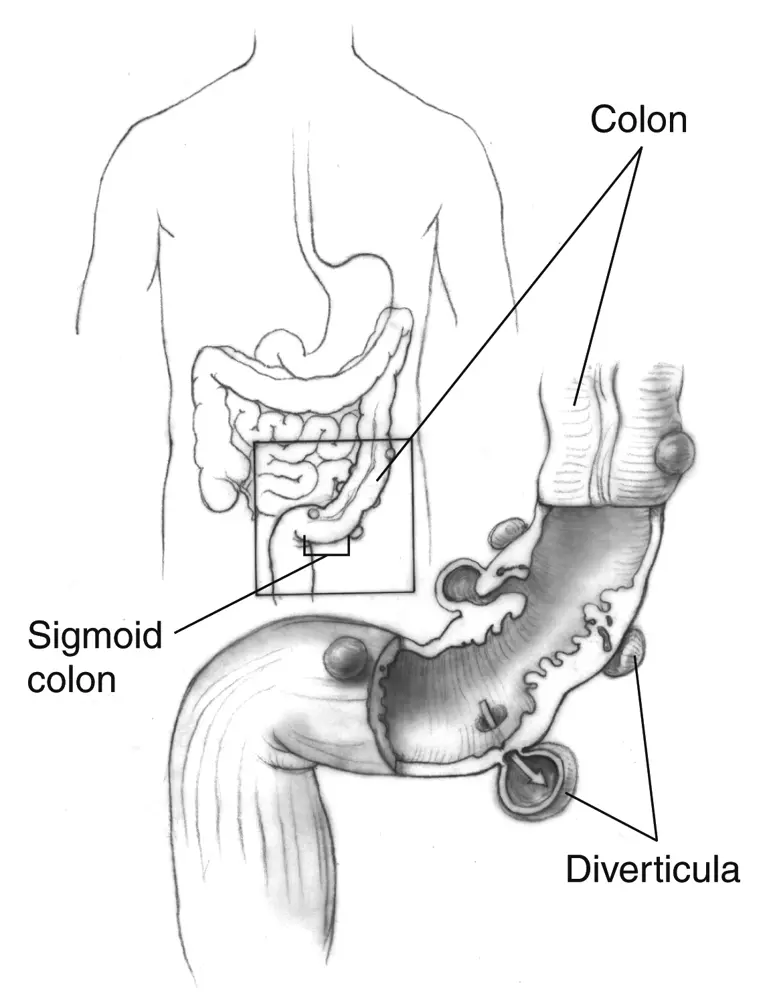
Diverticulitis describes inflammation and often infection within a diverticulum, most commonly in the sigmoid colon. The condition often begins abruptly with steady, localized pain, typically in the lower-left abdomen in Western populations. Patients may also experience fever, nausea, and either constipation or diarrhea. The same person can have symptom-free periods between discrete “attacks,” which vary in intensity from mild to severe.
Diverticulosis vs. diverticulitis: the key distinction
Diverticulosis means the pouches exist, often causing no symptoms and discovered incidentally during colonoscopy or imaging. Diverticulitis means one or more of those pouches have become inflamed/infected, producing clinical symptoms and sometimes complications. The umbrella term diverticular disease is used when diverticula cause symptoms or problems, including chronic abdominal discomfort, bleeding, or acute diverticulitis episodes.
Epidemiology: who tends to get diverticulitis?
Diverticulosis is very common and increases with age; a large share of adults over 50 have diverticula, and prevalence climbs further in the eighth decade. Only a minority progress to diverticulitis, but the absolute number of cases is high, driving substantial outpatient visits and hospitalizations each year. In recent decades, clinicians have observed more cases among adults under 40, possibly reflecting diet and lifestyle shifts. Among those under 50, men appear more frequently affected; after 50, women predominate.
Ethnic and geographic patterns vary: in Western populations, sigmoid (left-sided) disease dominates; in some Asian cohorts, right-sided diverticulitis is more common, shifting the typical pain location to the right lower quadrant. These differences affect how clinicians interpret symptoms and imaging findings in diverse populations.
Causes & pathophysiology: how attacks begin
Diverticula generally arise at points where blood vessels penetrate the colonic wall. Over time, increased intraluminal pressure—linked to low-fiber diets and constipation—can push the mucosal layer out through these weak points. An acute diverticulitis flare may be triggered when a pouch neck becomes obstructed (for example by a fecalith), allowing bacteria to proliferate and irritate the wall. Micro-perforation and contained infection can follow, sometimes evolving into a localized abscess.
Modern research highlights a role for the gut microbiome and the host inflammatory response. Some flares may occur without a discrete obstruction, driven by mucosal injury, local immune activation, and shifts in microbial communities. Diets high in red meat and low in fiber may promote inflammatory tone and alter microbial metabolites, whereas plant-rich patterns are associated with lower risk.
Risk factors you can address (and those you can’t)
Risk accumulates with age, but modifiable factors matter: low dietary fiber, high red-meat intake, sedentary behavior, obesity, tobacco use, and regular NSAID or steroid exposure. Family history and possible vitamin D insufficiency also correlate with risk in some studies. Addressing lifestyle drivers can lower future flare risk even after an initial episode.
Risk Factors at a Glance
Quick-reference cards summarizing how each factor contributes and what typically helps.
Modifiable & Nonmodifiable Drivers
Use these levers between flares to reduce recurrence risk and improve comfort.
Common symptoms (and when to seek urgent care)
The hallmark symptom is steady lower-left abdominal pain that often starts suddenly and worsens over hours. Many patients develop fever and chills, bloating, and bowel-habit changes. Nausea may occur; some people note urinary frequency/urgency when inflamed sigmoid colon irritates the bladder. Right-sided pain is more common in some Asian populations.
Symptoms & What They Suggest
Recognize patterns early and know when to escalate care.
Quick Clinical Cues
Worsening pain, high fever, rigid abdomen, or fainting = emergency care.
How diverticulitis is diagnosed
Clinicians synthesize the history and exam with labs and imaging. Blood tests frequently show elevated white blood cells and CRP, though these are nonspecific. The reference imaging test is a contrast-enhanced CT scan of abdomen/pelvis, which can confirm inflamed diverticula, colonic wall thickening, fat stranding, and complications such as abscess, fistula, or free air.
Endoscopy (colonoscopy or sigmoidoscopy) is generally avoided during an acute attack because of perforation risk. After recovery—often around 6–8 weeks later—clinicians may recommend colonoscopy to rule out alternative diagnoses (for example, malignancy) and to update routine colorectal screening if due.
Hinchey staging and why it matters
On CT, acute diverticulitis is often framed as uncomplicated (localized inflammation without abscess, perforation, obstruction, or fistula) or complicated (one or more of those present). The modified Hinchey classification adds nuance: I–II represent pericolic or distant abscess; III indicates generalized purulent peritonitis; IV indicates fecal peritonitis. Staging guides whether conservative care, image-guided drainage, or emergency surgery is most appropriate.
Hinchey Stages (CT-Based)
Use these quick cards to orient treatment strategy.
Stage I–IV Overview
Escalate promptly with systemic toxicity, diffuse peritonitis, or failure of conservative therapy.
Treatment: conservative, interventional, and surgical
For mild, uncomplicated diverticulitis, many patients can be managed at home. Early measures commonly include a brief period of clear liquids or a gentle low-fiber diet, rest, hydration, and analgesia with acetaminophen rather than NSAIDs. Some clinicians prescribe oral antibiotics; others follow a selective approach in immunocompetent adults with mild disease, provided close follow-up is feasible.
Hospital care is indicated for severe pain, high fever, inability to tolerate oral intake, or complications. Management typically includes IV fluids, IV broad-spectrum antibiotics, and bowel rest. If CT demonstrates an abscess (especially ≥4–5 cm), interventional radiology can often place a percutaneous drain to control sepsis and avoid emergency surgery. Failure to improve, or evidence of perforation, fistula, or obstruction, prompts surgical consultation.
Surgical strategies depend on acuity. In unstable emergencies with diffuse peritonitis, surgeons may perform resection with temporary colostomy (e.g., Hartmann’s procedure). In more controlled settings, minimally invasive sigmoid resection with primary anastomosis is common, often avoiding permanent stoma. Elective surgery is individualized rather than triggered by a fixed number of episodes, focusing on quality-of-life impact, complication history, and patient preference.
Treatment Scenarios: Quick Cards
Match the scenario to typical first steps. Always individualize with your clinician.
From Mild Flare to Emergency
Escalate care if pain, fever, or intolerance to oral intake persists or worsens.
How serious is diverticulitis?
Most uncomplicated episodes resolve within days to a week with appropriate care. Serious risk rises with complications such as abscess, perforation with peritonitis, obstruction, or fistulae (for example, a colovesical fistula causing recurrent UTIs). Rapid assessment and treatment dramatically lower the chance of severe outcomes, and overall life expectancy is generally unaffected in successfully treated cases.
Diet during flare & recovery
Eating patterns change across phases. In the first day or two of a mild flare, clinicians often suggest clear liquids or very low-fiber, easily digested foods to reduce stool bulk and mechanical stress. As pain and fever settle, patients typically advance to soft, low-fiber choices, then gradually return to a balanced, higher-fiber pattern over about a week (or per clinician guidance).
Diverticulitis Diet Examples: Acute Flare vs. Recovery Day
The right foods depend on the phase you are in. Below are two sample daily plans:
one for the first 24–48 hours of a mild flare (bowel rest focus), and one for a
post-recovery day emphasizing gradual return to fiber. Use these as educational
examples—follow your clinician’s instructions for your specific case.
Acute Flare (First 24–48 Hours—Gentle & Low Fiber)
Goal: bowel rest, hydration, and symptom monitoring; transition as symptoms settle.
Recovery Day (Gradual Return to Balanced Fiber)
Goal: gradually reintroduce fiber and textures without provoking symptoms.
A long-standing myth suggested avoiding nuts, seeds, corn, and popcorn with diverticular disease. Current evidence does not support this restriction; these foods do not increase diverticulitis risk and can contribute useful fiber in remission phases.
Prevention: habits that support colon health
Between attacks, a fiber-forward pattern is associated with fewer symptoms and lower risk of future flares. Aim to build fiber from fruits, vegetables, legumes, and whole grains, increasing slowly to minimize gas and bloating. Hydration helps fiber do its job; without fluid, stool can remain hard.
Regular physical activity improves bowel motility and supports weight management, both linked to lower risk. Limiting red and processed meats and prioritizing plant proteins and fish may further reduce inflammatory tone. If you use NSAIDs frequently, ask your clinician about alternatives, as NSAIDs are associated with bleeding and diverticular complications.
Some clinicians recommend fiber supplements (e.g., psyllium) if dietary fiber is consistently low. Probiotic foods or supplements are being studied; while evidence is mixed, a few small trials suggest potential benefit for recurrence reduction in selected patients. Discuss whether these are suitable for you.
Long-term outlook & recurrence
Most people recover fully from an acute episode. Recurrence risk exists—roughly a minority will experience a later flare—but many never do. A small subset develop chronic smoldering symptoms or complications (such as strictures, fistulas) that may ultimately benefit from elective surgery. Early follow-up, lifestyle measures, and adherence to clinician guidance improve long-term comfort and function.
Emerging guidelines: antibiotics & surgery are individualized
Randomized trials and society guidance have shifted practice away from “antibiotics for everyone” in mild, uncomplicated cases toward selective use in immunocompetent adults with reliable follow-up. This approach aims to reduce unnecessary exposure and resistance while preserving safety. Close monitoring is essential; any deterioration should prompt antibiotics and reassessment.
The historical “operate after two attacks” rule has been replaced by individualized decisions. Indications for surgery now center on complication profiles (e.g., fistula, obstruction), severity and frequency of flares, patient values, and overall risk. Minimally invasive techniques enable faster recovery in many elective cases.
Myths vs. Evidence
Clear up common misconceptions with practical takeaways.
What to Know
Evidence evolves—review your plan at follow-up visits, especially after any flare.
Frequently Asked Questions about Diverticulitis
How long does a mild diverticulitis flare usually last?
Many uncomplicated flares begin improving within 48–72 hours after supportive care (and antibiotics when used), with most people feeling much better by a week. Recovery timelines vary with severity and comorbidities; follow your clinician’s directions for diet progression and activity.
Do women have different symptoms?
Core symptoms are similar across sexes. In reproductive-age women, lower-abdominal pain may overlap with gynecologic conditions, so clinicians consider ovarian or uterine causes in the differential. Digestive changes plus fever tilt suspicion toward diverticulitis, but imaging clarifies the diagnosis.
When should I go to the ER?
Seek urgent care for severe or worsening abdominal pain, high fever, a rigid or distended abdomen, inability to keep fluids down, fainting, or signs of widespread infection. Bright red blood in stool also warrants prompt evaluation.
Do I need a colonoscopy after my first attack?
If you haven’t had age-appropriate colorectal screening or if imaging is atypical, clinicians often recommend colonoscopy several weeks after recovery to rule out other pathology. The timing is individualized; many aim for ~6–8 weeks post-flare.
Can probiotics help prevent recurrence?
Research is ongoing. Small trials suggest possible benefit in select patients, but findings are mixed and not yet definitive. If you’re considering probiotics, discuss strains, dosing, and duration with your clinician to ensure safety and realistic expectations.
Precautions
This educational article does not replace personalized medical advice. People with immune compromise, significant comorbidities, severe pain or fever, repeated vomiting, suspected bleeding, or pregnancy should seek clinician guidance promptly. Medication choices (including antibiotics and pain control) and diet advancement should be individualized.
Content reflects summaries from reputable clinical sources and is intended for general information. Always follow the plan provided by your healthcare professional.
